
Our refined molecular and cellular taxonomy can provide a framework for stratification in clinical trials and serve as a roadmap for therapeutic targeting of specific cellular phenotypes and multicellular interactions. Integrating spatial and cellular profiles revealed three multicellular communities with distinct contributions from malignant, fibroblast and immune subtypes: classical, squamoid-basaloid and treatment enriched. 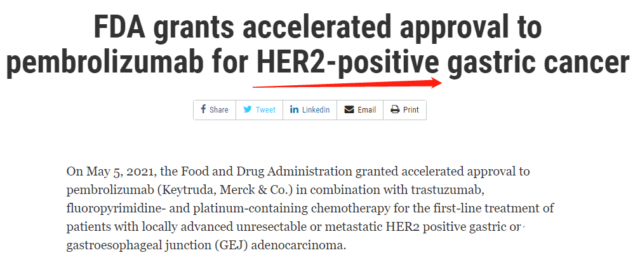
We uncovered recurrent expression programs across malignant cells and fibroblasts, including a newly identified neural-like progenitor malignant cell program that was enriched after chemotherapy and radiotherapy and associated with poor prognosis in independent cohorts. Here, we construct a high-resolution molecular landscape of the cellular subtypes and spatial communities that compose PDAC using single-nucleus RNA sequencing and whole-transcriptome digital spatial profiling (DSP) of 43 primary PDAC tumor specimens that either received neoadjuvant therapy or were treatment naive. Molecular stratification in pancreatic cancer remains rudimentary and does not yet inform clinical management or therapeutic development. Pancreatic ductal adenocarcinoma (PDAC) is a highly lethal and treatment-refractory cancer. Three recently published studies used this technology for large-scale sequencing of TPA, which enabled the deep exploration of modern syphilis epidemics1,2,3. Hybridization capture using DNA or RNA probes designed based on TPA genomes has become the most popular and efficient technique for this targeted genome sequencing. During the past 6 years, several culture-independent enrichment techniques have partially addressed the problem of low bacterial loads against a background of highly abundant host DNA in clinical samples, and whole-genome sequencing has become possible directly from patient samples. Difficulties in culturing TPA in vitro from clinical samples have limited our understanding of the global diversity of this host-adapted pathogen, which has further hampered vaccine development. pallidum (TPA), has spurred the need for a vaccine. The worldwide increase in syphilis, caused by the sexually transmitted spirochete Treponema pallidum subsp. Planned enrollment is approximately 692 patients.This Genome Watch highlights how population genomic strategies can aid in the development of an effective syphilis vaccine. Patients will be followed up for survival. 
Adverse events are graded per NCI CTCAE v4.0 and will be monitored for 30 or 90 days after treatment. Secondary endpoints are objective response rate, duration of response, and safety and tolerability. Primary endpoints are progression-free survival and overall survival. Investigator choice chemotherapy will include day 1 cisplatin 80 mg/m 2 IV and 5-fluorouracil 800 mg/m 2 /day IV (days 1-5) or oxaliplatin 130 mg/ m2 IV and capecitabine 1000 mg/m 2 BID days 1-14 (Q3W). Patients will be randomly assigned 1:1 to receive chemotherapy with pembrolizumab 200 mg IV flat dose or placebo with trastuzumab 6 mg/kg (after 8 mg/kg load) Q3W up to 2 years or until intolerable toxicity or disease progression. Methods: Key eligibility criteria are age ≥18 years previously untreated unresectable or metastatic HER2+(centrally confirmed IHC 3+ or IHC 2+/ISH >2.0) G/GEJ adenocarcinoma life expectancy >6 months with RECIST v1.1 measurable disease adequate organ function and performance status. KEYNOTE 811 (, NCT03615326), a global, multicenter, randomized, placebo-controlled, phase 3 study, is underway. 
This phase 2 study in HER2+ mG/GEJc demonstrated the safety and preliminary efficacy of trastuzumab/pembrolizumab/chemotherapy the overall response rate was 87%, and the disease control rate was 100% (Janjigian YY, ASCO GI 2019). We hypothesize that combination anti-PD-1 and anti-HER2 therapy will result in T-cell activation, augment ADCC, and potentiate antitumor immune response in HER2+ patients. Introduction: Combination therapy with the anti-HER2 antibody trastuzumab with fluoropyrimidine and platinum is the current standard for patients with HER2+ metastatic gastric or gastroesophageal junction cancer (mG/GEJc).
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
